Barocaloric thermal batteries: Concept and realization. Credit: Institute of Metal Research
A Chinese research team has developed a new concept for extracting thermal energy from low-temperature waste heat sources and reusing it on demand simply by controlling the pressure.
Heat production accounts for more than 50% of the world's final energy consumption and analysis of waste heat potential shows that 72% of the world's primary energy consumption is lost after conversion, mainly in the form of heat. It is also responsible for more than 30% of global greenhouse gas emissions.
Against this background, researchers led by Prof. Li Bing from the Institute of Metal Research of the Chinese Academy of Sciences have proposed and realized a new concept—barocaloric thermal batteries based on the unique inverse barocaloric effect.
The study was published in Science Advances.
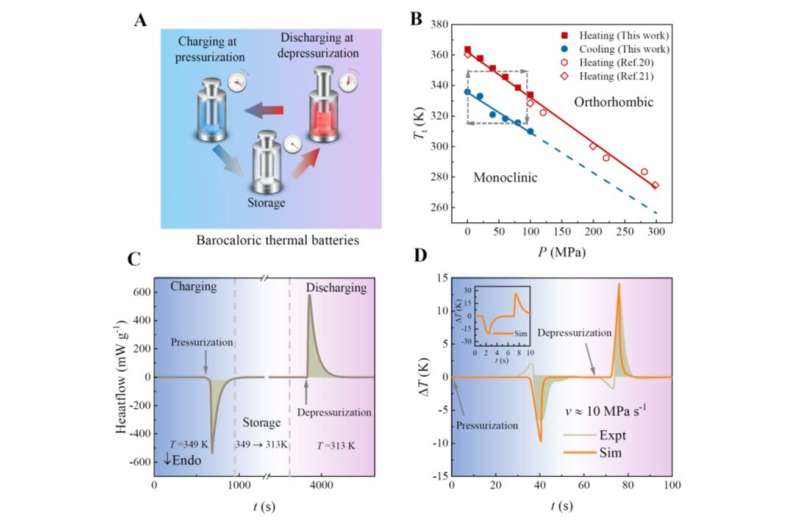
An inverse barocaloric effect is characterized by a pressure-induced endothermic response, in sharp contrast to a normal barocaloric effect where pressurization leads to an exothermic response. "A barocaloric thermal battery cycle consists of three steps, including thermal charging upon pressurization, storage with pressure, and thermal discharging upon depressurization," said Prof. Li, corresponding author of the study.
The barocaloric thermal battery was materialized in ammonium thiocyanate (NH4SCN). Discharge was manifested as the heat of 43 J g-1 or a temperature rise of about 15 K. The heat released was 11 times greater than the mechanical energy input.
To understand the physical origin of the unique inverse barocaloric effect, the working material NH4SCN has been well characterized using synchrotron X-ray and neutron scattering techniques. It undergoes a crystal structural phase transition from a monoclinic to an orthorhombic phase at 363 K, accompanied by a volumetric negative thermal expansion of ~5% and entropy changes of about 128 J kg-1 K-1.
This transition is easily driven by pressure as low as 40 MPa, and it is the first inverse barocaloric system with entropy changes greater than 100 J kg-1K-1. Pressure-dependent neutron scattering and molecular dynamics simulations showed that the transverse vibrations of SCN¯ anions are enhanced by pressure and the hydrogen bonds that form the long-range order are then weakened.
As a result, the system becomes disordered in response to external pressure and thus the material absorbs heat from the environment.
As an emerging solution for manipulating heat, barocaloric thermal batteries are expected to play an active role in a variety of applications such as low-temperature industrial waste heat harvesting and reuse, solid-state refrigeration heat transfer systems, smart grids, and residential heat management.