Researchers at North Carolina State University, USA, have developed a new technique for extracting hydrogen gas from liquid carriers.
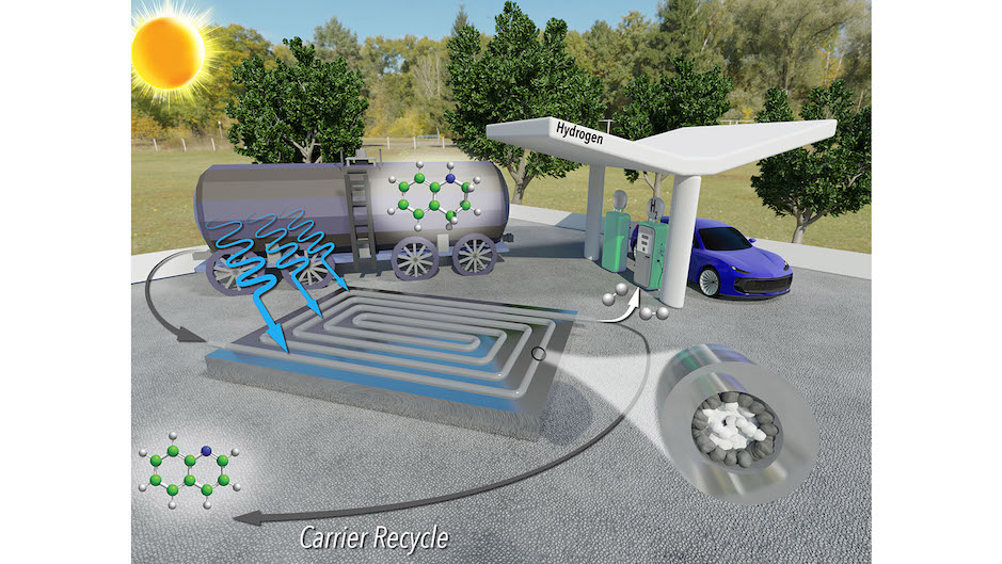
Image credit: NC State University
According to the team, their new method is faster, less expensive and more energy efficient than previous approaches.
Milad Abolhasani, associate professor of chemical and biomolecular engineering at NC State and a corresponding author of a paper on the technique, said that there are technical obstacles that need to be overcome before hydrogen can be viewed as a practical sustainable energy alternative.
Hydrogen refuelling stations could be located at existing petrol stations, taking advantage of existing infrastructure, but transporting hydrogen is dangerous, so it needs to be transported by a liquid carrier. A key obstacle for this strategy is that extracting hydrogen from the liquid carrier at destination sites, such as fuelling stations, is energy intensive and costly.
“One of the big obstacles to the adoption of a hydrogen economy is the cost of storage and transportation,” Abolhasani said. “Previous research has shown that it is possible to use photocatalysts to release hydrogen gas from a liquid carrier using only sunlight.
“However, existing techniques for doing this were laborious, time consuming and required a significant amount of rhodium – a metal that is very expensive.”
Now, according to the paper’s first author Malek Ibrahim, a former postdoctoral researcher at NC State, the team has developed a technique that applies a reusable photocatalyst and sunlight to extract hydrogen from its liquid carrier more quickly and using less rhodium. He added that the only by-products are hydrogen gas and the liquid carrier itself, which can be reused repeatedly.
Researchers said that key to the new technique’s success is that it is a continuous-flow reactor. The reactor resembles a thin, clear tube packed with sand. The ‘sand’ consists of micron-scale grains of titanium oxide, many of which are coated with rhodium.
The rhodium-coated particles line the tube’s outer part, where sunlight can reach them. These particles are photoreactive catalysts that, in the presence of sunlight, react with the liquid carrier to release hydrogen molecules as a gas. The team said they precisely engineered the system so that only the outer grains of titanium oxide are coated with rhodium.
“In a conventional batch reactor, 99 per cent of the photocatalyst is titanium oxide and one per cent is rhodium,” said Abolhasani. “In our continuous flow reactor, we only need to use 0.025 per cent rhodium, which makes a big difference in the final cost. A single gram of rhodium costs more than $500.”
In their prototype reactor, researchers reported achieving a 99 per cent yield in three hours.
“That’s eight times faster than conventional batch reactors, which take 24 hours to reach 99 per cent yield,” Ibrahim said. “And the system should be easy to scale up or scale out to allow for catalyst reuse on commercial scale — you can simply make the tube longer or merge multiple tubes running in parallel.”
The flow system can reportedly run continuously for up to 72 hours before its efficiency decreases. At this point, the catalyst can be ‘regenerated’ without removing it from the reactor, a simple cleaning process that takes about six hours, researchers said. The system can then be restarted and run at full efficiency for another 72 hours.
NC State has filed a provisional patent for the technology.